Warm air can have a higher moisture content than cooler air.
True. Air at 80 deg F and 50% relatively humidity contains more moisture by mass per volume of air than air at 40 deg F and 50% humidity.
At 100°C the moisture can be 100%. At 0°C the moisture is 0%. In between is relative humidity.
False. Don't feel bad, Nightmare, as this is a common misperception.
The relative humidity of an air-water mixture is defined as the ratio of the partial pressure of water vapor (H2O) in the mixture to the equilibrium vapor pressure of water at a given temperature.
In a mixture of gases, each gas has a partial pressure which is the hypothetical pressure of that gas if it alone occupied the volume of the mixture at the same temperature.
Vapor pressure or equilibrium vapour pressure is defined as the pressure exerted by a vapor in thermodynamic equilibrium with its condensed phases (solid or liquid) at a given temperature in a closed system. The equilibrium vapor pressure is an indication of a liquid's evaporation rate.
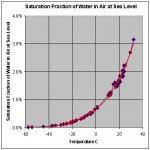
As the graph clearly shows, atmosphere at -20 deg C (-4 deg F, about what is it outside right now), can still hold plenty of moisture without that moisture precipitating out.
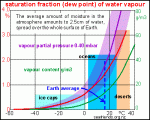
In fact, let's take a closer look at the actual weather conditions at the U.S. Air Force Academy right now:
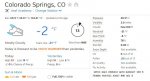
You you can see, it's -2 deg F (-18.89 deg C) outside right now. Yet the relative humidity is 78%, not 0%, as you claimed it would be, and the dew point is -8 deg F (-22.22 deg C). At a given temperature but independent of barometric pressure, the dew point is a consequence of the absolute humidity, the mass of water per unit volume of air.
At the dew point the moisture condenses on cooler surfaces. Heaters are intended to stabilize the temperature above the dew point. The humidity is still there, still causing rust.
True. Rust is an iron oxide, usually red oxide formed by the redox reaction of iron and oxygen in the presence of water or air moisture. Rust consists of hydrated iron(III) oxides Fe2O3·nH2O and iron(III) oxide-hydroxide (FeO(OH), Fe(OH)3).
Like all chemical reactions, corrosion rate is temperature dependent. A higher temperature causes a higher corrosion rate.
False. Rust in the oxidation of a metal, in particular, iron. It's an exothermic reaction, meaning it gives off heat. Applying heat to endothermic reactions speeds them up. Applying heat to exothermic reactions slows them down, and at some point, actually reverses the reactions. This is why the exothermic "hand warmers" are reenergized by plopping them into boiling water for a few minutes.
Furthermore, at lower temperatures, the relative humidity is higher, thereby being more readily available to interact with iron and oxygen to cause rust.
So, how do we prevent rust on our firearm treasures?
As with other metals, like aluminium, a tightly adhering oxide coating, a passivation layer, protects the bulk iron from further oxidation. The conversion of the passivating ferrous oxide layer to rust results from the combined action of two agents, usually oxygen and water.
This is why bluing works. It's a technique that can provide limited resistance to rusting for small steel items, such as firearms; for it to be successful, a water-displacing oil is rubbed onto the blued steel and other steel.
Thus, if you have a blued firearm, you need to ensure it's coated in a water-displacing oil. As for which is best, the debate continues to run rampant, despite a thousand years of data.
However, I know of no finer essay on this than
Day at the Range's Comprehensive Corrosion Test and Comparison of 46 Different Products.
See water h-s Mollier Diagram of enthalpy-entropy.
The Mollier Diagram plots lines of constant pressure, constant temperature, and volume, and the abscissa and ordinate represent entropy and heat. It's nearly identical to a psychometric chart, which represents the thermodynamic parameters of moist air at constant temperature.
According to the Mollier Diagram (as well as the psychometric chart), water vapor depositing on room temperature metal transfers heat from the metal due to the latent heat of evaporation (enthalpy of evaporation) caused by a phase change from gas to liquid. Wet skin in dry air cools as the water evaporates. When water vapor condenses on metal, it warms it. Thus, the cooler the metal, the more rapid the condensation.
The thing about rust, however, is that it occurs above dew point, too. In actuality, given a piece of iron like a steel handgun, whose temperature is above the dew point, no condensation will accumulate. However, this is simply because the condensation is evaporating faster than water vapor is condensing. At the microscopic level, water vapor contacts the metal all the time. It just doesn't stick around very long, unless the water molecule and an oxygen molecule happen to bump into an unbound iron molecule at the same time. When they do, the iron gives off an electron to the oxygen molecule, aided by the water molecule, which oxidizes the iron into rust, releasing hydrogen gas.
Bottom Line:
1. Guns rust faster in cooler temps than they do in warmer temps.
2. Guns rust faster in higher relative humidity than they do in drier climes.
3. The presence of chloride ions, such as occurs when the surface is contaminated with salt. The iron doesn't care whether that's from seawater or sweat from your hands, so wash, rinse, and dry your hands thoroughly before cleaning your firearm. Alternatively, just wear gloves made of Buna-N, Perbunan, acrylonitrile butadiene rubber (the purple, hypoallergenic surgical gloves).
4. Whether your firearm is blued or not, keeping it coated with
a quality, moisture-dispersing oil will significantly impede the oxidation process.
5. Don't believe everything you read on the Internet. If you're unfamiliar with the chemistry or thermodynamics yourself, the old watchword applies: Don't try this at home!